Research
Highlights...
|
 |
Number 283 |
March 30, 2009 |
|
Visualizing every breath you take
Airflow patterns in the lung not only determine how well you breathe but also how
inhaled materials such as airborne pollutants or aerosolized drugs are distributed inside your
body. Researchers from the Department of Energy’s Pacific Northwest National Laboratory and the University of Utah have pioneered a magnetic resonance imaging method for visualizing inhaled airflow patterns. Developed at DOE’s EMSL, a national scientific user facility, the method uses hyperpolarized helium-3 gas as an inert tracer for visualizing inhaled air speed
and direction at each location within the complex, three-dimensional airways of pulmonary anatomy. This method provides an opportunity to assess the subtle effects of inhaled pollutants and pesticides. Also, it allows scientists to visualize the electrochemistry in operating fuel cells to improve efficiency and reliability.
[Kristin Manke, 509.372.6011,
kristin.manke@pnl.gov] |
|
New breast cancer imager/biopsy system comes online
With help from DOE's Jefferson Lab, a system for detecting and guiding the biopsies of suspicious breast tumors is now in clinical testing at West Virginia University. The first patient was scanned with the device on Feb. 6; and by mid-March, a total of four patients had been scanned. The technology is built on traditional PET imaging, where a drug with a radiation-emitting component is injected into a patient and is then imaged. If a suspected lesion is found, a biopsy is performed with a computer and human-controlled robotic arm. The PEM/PET system was designed and constructed by scientists and engineers from WVU, JLab, the University of Washington and the University of Maryland School of Medicine.
[Kandice Carter, 757.269.7263,
kcarter@jlab.org] |
|
Carbon boosts hydrogen production
One of the keys to making a hydrogen-powered future feasible is developing environmentally and economically sound methods of producing large quantities of hydrogen. Biological hydrogen production by cyanobacteria, also called blue-green algae, is a highly attractive option, using a renewable resource requiring only water, sunlight, air, and trace mineral salts. A team of Savannah River National Laboratory researchers is studying how the relationships between blue-green algae and its environment, particularly nutrients and other bacteria, affect its ability to produce hydrogen that could be collected and used for the nation’s energy needs. Results indicate that a carbon “boost” may increase the hydrogen production capacity of many strains. SRNL examined 10-12 diverse cyanobacterial strains and found that glucose stimulated hydrogen production rates and yield in the majority of strains—as much as a 40-fold increase in yields in some strains. Other carbon sources were also found to increase cyanobacterial hydrogen production. These results support the idea that organic rich waste streams from certain industrial processes could be used to stimulate photobiological hydrogen production.
[Angeline French, 803.725.2854, angeline.french@srnl.doe.gov] |
Desalination of saline and brackish water becoming more affordable
 |
| Sandia researcher Susan Altman conducts research to minimize biofouling on reverse osmosis membranes used for desalination in her laboratory (Photo by Randy Montoya) |
Treating brackish water for human consumption “can be done and be done affordably” in various parts of the U.S., according to Sandia National Laboratories water researcher Mike Hightower. While it used to cost 50 cents per thousand gallons of water to supply freshwater, it now costs $3 to $4 per thousand gallons of water, due largely to the fact that freshwater near cities is generally already being used and utilities frequently have to pump water long distances, he said. Meanwhile, the cost of treating saline and brackish water has decreased to $2 to $3 per thousand gallons for water treated at the ocean and $4 to $6 per thousand gallons for inland treatment.
[Julie Hall, 505.284.7761,
jchall@sandia.gov] |
Novel fluid-like nanoscale material growth
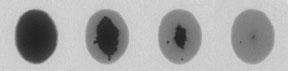 |
| Lead atoms in liquid-like motion across a silicon substrate |
When physicists at DOE’s Ames Laboratory examined how lead atoms self assemble into nanoscale stacks, or islands, on a silicon surface, they made two surprising discoveries.
“One big surprise was that the atoms were moving a lot at such a low temperature,” said senior physicist Michael C. Tringides, who led the research effort. “The other surprise was that the atoms weren’t moving randomly like individual atoms as expected. Instead, the whole layer of lead atoms was moving like a liquid.” The unexpected observations of fluid-like motion help explain the unique fast and uniform growth behavior of lead-on-silicon islands. And, the findings offer promising possibilities for the next generation of nanosized transitors or novel nanomaterials, since industrial applications of nanogrowth require fast and easily reproducible processes.
[Breehan Gerleman Lucchesi, 515.294.9750, breehan@ameslab.gov] |
|
|
LANL’s Kathy Prestridge
explores fluid instabilities
Kathy Prestridge sees her work at DOE’s Los Alamos National Laboratory as an exciting learning experience. “There is a lot of interesting science going on here, and I enjoy working with a technically diverse group of people,” she says.
Prestridge is the principal investigator in LANL’s Gas Shock Tube Project. “We study fluid instabilities and turbulent mixing in shock-driven flows, and our team has made the first simultaneous velocity and density field measurements in these flows,” she says. “The main diagnostics that we use are Particle Image Velocimetry (PIV) and Planar Laser Induced Fluorescence (PLIF) to get the velocity and density fields instantaneously and in two dimensions. We can then take averages over multiple experiments and calculate the mean and fluctuating density and velocity quantities.”
These quantities have never before been measured experimentally, and this year, Prestridge and her team are working on developing a stereo PIV system to measure all three components of the velocity field. The experimental data are valuable for understanding the nature of mixing, the impact of initial conditions on unsteady turbulence, and to aid the validation and calibration of turbulence mixing models used in simulations, she says.
The group also is involved in a Laboratory Directed Research and Development–Directed Research (LDRD-DR) project, "Turbulence by Design," that studies the impact of initial conditions on the evolution to turbulence of shock-driven flows. It incorporates theory, the group’s experiments, and numerical modeling to understand and potentially control turbulence in Rayleigh-Taylor and Richtmyer-Meshkov flows.
Prestridge holds a bachelor's degree in aerospace engineering from Princeton University and a doctorate in applied mechanics from the University of California, San Diego. She won the Postdoctoral Publication Prize in Experimental Sciences in 2000 and was named a LANL Star in 2008 for her technical work and her achievements as a mentor, project leader, and deputy group leader.—Tatjana K. Rosev
Submitted by DOE's Los Alamos National Laboratory
|
|




