- Number 315 |
- July 6, 2010
-
Energy saving air conditioner conquers all climates
 DOE’s National Renewable Energy Laboratory has invented a new air conditioning process with the potential of using 50 percent to 90 percent less energy than today’s top-of-the-line units. It uses membranes, evaporative cooling and liquid desiccants in a way that has never been done before in the centuries-old science of removing heat from the air.
DOE’s National Renewable Energy Laboratory has invented a new air conditioning process with the potential of using 50 percent to 90 percent less energy than today’s top-of-the-line units. It uses membranes, evaporative cooling and liquid desiccants in a way that has never been done before in the centuries-old science of removing heat from the air.
-
New energy research under the microscope
The findings point the way toward future work that could greatly improve catalyst efficiency in a variety of processes that are crucial to the world’s energy security, such as petroleum catalysis and catalyst-based nanomaterial growth for next-generation rechargeable batteries. Scientists can now peer into the inner workings of catalyst nanoparticles 3,000 times smaller than a human hair within nanoseconds.
Scientists can now peer into the inner workings of catalyst nanoparticles 3,000 times smaller than a human hair within nanoseconds.
-
Evaluating natural gas hydrate occurrences around the world
 Working on research ships around the world and Arctic drilling rigs, as well as in the laboratory, researchers in NETL’s Office of Research and Development perform integrated studies that may hold the secret to greatly increasing our Nation’s natural gas supply. Hydrate is an informal term commonly used to describe substances in which molecules of water form a cage that encloses, without chemically bonding with, appropriately-sized molecules of another material (in this case, methane).
Working on research ships around the world and Arctic drilling rigs, as well as in the laboratory, researchers in NETL’s Office of Research and Development perform integrated studies that may hold the secret to greatly increasing our Nation’s natural gas supply. Hydrate is an informal term commonly used to describe substances in which molecules of water form a cage that encloses, without chemically bonding with, appropriately-sized molecules of another material (in this case, methane). -
Light, sound, action: The plasmonic promise of graphene
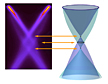 Scientists working at the Advanced Light Source (ALS) at DOE’s Lawrence Berkeley National Laboratory have discovered striking new details about the electronic structure of graphene, crystalline sheets of carbon just one atom thick. An international team led by Aaron Bostwick and Eli Rotenberg of the ALS found that composite particles called plasmarons play a vital role in determining graphene’s properties.
Scientists working at the Advanced Light Source (ALS) at DOE’s Lawrence Berkeley National Laboratory have discovered striking new details about the electronic structure of graphene, crystalline sheets of carbon just one atom thick. An international team led by Aaron Bostwick and Eli Rotenberg of the ALS found that composite particles called plasmarons play a vital role in determining graphene’s properties.
-
Opposites attract and inspire electrocatalyst
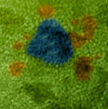 Tiny gold particles surround themselves with even smaller platinum bits, creating a complex structure that could turn a common preservative, formic acid, into electricity in a fuel cell, according to scientists from China's Harbin Institute of Technology and DOE’s Pacific Northwest National Laboratory. The team used a novel electrostatic self-assembly method to create platinum-surrounded gold nanomaterial.
Tiny gold particles surround themselves with even smaller platinum bits, creating a complex structure that could turn a common preservative, formic acid, into electricity in a fuel cell, according to scientists from China's Harbin Institute of Technology and DOE’s Pacific Northwest National Laboratory. The team used a novel electrostatic self-assembly method to create platinum-surrounded gold nanomaterial.


