| Research
|
Dr. Carol Jantzen's professional interests have always seemed to focus on things that last a long time. Her first paying job as a child, the then-named Carol Fredericks helped her father collect and assemble rocks and minerals for educational kits led her into university studies in geochemistry and subsequently into materials science and engineering. Today, the Savannah River National Laboratory scientist and internationally recognized expert in glass and ceramics conducts research and development to ensure that the materials used for disposing of high-level radioactive waste will last for thousands of years. Dr. Jantzen came to DOE's SRNL in the 1980s as part of the laboratory team developing the processes to be used in the Savannah River Site's Defense Waste Processing Facility, the nation's first facility for converting high-level radioactive waste (HLW) into a stable glass form for permanent disposal, which began operation in 1994. Her contributions including the development of the statistical process control models for the process helped ensure a real solution for the decades-old problem of nuclear waste disposal. She has developed waste forms for a wide variety of high-level radioactive wastes and hazardous/mixed and low-level wastes in both the U.S. and Europe . Recently, she developed a new glass durability model that has the potential to increase the waste loading, or the amount of waste that can be incorporated into a HLW glass. At this fall's Materials Science & Technology conference, she will receive the Bleininger Award, recognizing excellence and lifelong achievements in the field of ceramics. As the first female president of the American Ceramic Society in 100 years, she helped found the society's Student Mentor Program and is committed to the encouragement of students and future ceramics professionals. Her ongoing work in developing solutions to important waste management challenges, combined with that dedication to future scientists, will ensure that her influence is felt for a very long time. Submitted by DOE's Savannah River National Laboratory |
|||||||||||||||||||||||||
|
See Lawrence Livermore's S&TR magazine.
|
New technology could fuel
|
 |
|---|
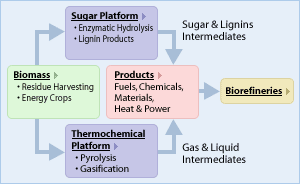
Researchers at DOE's Argonne National Laboratory and Archer Daniels Midland Co. (ADM) are developing a separative bioreactor that efficiently turns sugar from corn into valuable chemicals. The technology could help bio-based chemicals replace large amounts of petrochemicals, helping reduce U.S. dependence on foreign oil, benefiting rural economies and reducing greenhouse gas emissions.
The two-year joint research effort will evaluate and optimize the production of gluconic acid using the separative bioreactor. Eventually, the technology could extend beyond gluconic acid to the production of a variety of organic acids and polyols, which form the chemical building blocks for plastics, pharmaceuticals and other consumer products.
Gluconic acid is produced by the biochemical oxidation of glucose, a sugar found in corn starch. This reaction, facilitated by enzymes in fermentation broths, has been known for more than 100 years. The challenge is one of engineering how to process gluconic acid cheaply and cleanly enough to compete economically with petrochemicals.
The separative bioreactor could overcome technical and economic barriers to the production of gluconic acid, said Seth Snyder, the principal investigator at Argonne .
Argonne researchers have learned how to immobilize the enzyme that turns glucose into gluconic acid, and they have merged that capability with a separation process called electrodeionization, which uses electricity to remove even low concentrations of ions from a solution. Argonne researchers developed and patented an improved EDI resin wafer stack that won a 2002 R&D 100 Award. Funding for the stack research, which efficiently removes salt from high fructose corn syrup, was provided by DOE's Industrial Technology program.
Inside the separative bioreactor, enzymes convert a steady stream of glucose into gluconic acid. The gluconic acid ionizes and is immediately separated from the glucose solution by the EDI process.
The EDI separation eliminates a major problem in large-scale gluconic acid production the incompatibility of the enzyme and the product acid. The Argonne separative bioreactor operates continuously, using only electricity to separate the product. Snyder said the cost of the electricity is very, very small. It's well within our goals for the overall bioprocessing cost.
In the test-scale systems at Argonne , the process has been demonstrated effectively at speeds of about a gallon of glucose solution a day. A commercial-scale reactor would be several times larger, and hundreds of units would be stacked together to achieve industrial-scale output.
ADM, one of the largest producers of gluconic acid in the world, understands the economics of large-scale production, Snyder said, adding that economics not environmental considerations will determine the commercial success of any bio-based production process, although bio-based production does offer substantial environmental benefits.
Argonne 's research on separative bioreactors is part of a movement to develop biorefineries that turn raw biomass from crops, grasses and trees into electricity, transportation fuels and refined chemicals. This movement is supported by the U.S. Department of Energy Biomass Program in the Office of Energy Efficiency and Renewable Energy , which is funding Argonne 's research along with ADM.Submitted by DOE's Argonne National Laboratory



