| Research
|
|
|||||||||||||||||||||||||||
|
Check out the Office of Science's new Website.
|
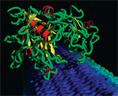
Simulations probe how enzymes crack cellulose
Harnessing the capabilities of one of the world's most powerful supercomputers, the largest biological simulation ever conducted at DOE's Oak Ridge National Laboratory is providing insights on converting cellulose from green plants into sugar. The supercomputer's ability to create a visualization of a million atoms promises to help researchers design more efficient methods for producing biofuels. Like humans, microbes depend on sugar for energy. Fungi and bacteria obtain sugar from cellulose, the glucose polymer that composes cell walls in trees and other plants on land and in the ocean. These microorganisms produce cellulases, enzymes that work together to extract glucose from cellulose and turn the sugar into energy. Computational biologists from ORNL and the National Renewable Energy Laboratory in Colorado, along with a researcher from Cornell University, are modeling bacterial and fungal cellulases in action at ORNL. Researchers "watch" these simulated enzymes attack digital cellulose strands, transfer a strand's sugar molecules to the enzyme's catalytic zone and chemically digest the sugar to provide the microbe with energy. The key to increasing the efficiency and lowering the cost of ethanol production using sugar from cellulose in treesand other biomass is to understand how cellulases degrade cellulose. Such understanding may lead to genetic engineering of the degradation mechanisms, speeding the biofuel production process. John Brady of Cornell developed the original model of a cellulase molecule processing cellulose fibers under an optical microscope. Led by NREL's Mike Himmel, other collaborators include Ed Uberbacher and Phil LoCascio, researchers in ORNL's Genome Analysis and Systems Modeling Group, and Pavan K. Ghattyvenkatakrishna, a University of Tennessee graduate student who works in the GASM Group. "We are studying the dynamics of cellulase at work on cellulose," says Uberbacher. "The model's cellulose resembles wood under a microscope. The individual strands are sugars strung together as a long polymer chain. The cellulase enzyme comes down and pulls a strand from the bundle forming the glucose polymer. The cellulase feeds the sugar fiber up to another domain of the enzyme that catalyzes the removal of the six-carbon sugar from the fiber. The process is a crucial step in making ethanol fuel from biomass." The goals of the project include understanding how the cellulase enzyme functions, how it recognizes cellulose strands and how the chemistry is accomplished inside the enzyme. The group also hopes to determine what the rate-limiting steps are that might be genetically engineered to make cellulase more efficient at degrading cellulose into glucose. Computational biologists hope to explore the diversity among cellulases and figure out why some cellulases work better at extremely high temperatures while others prefer lower temperatures. One question researchers hope to address is how fungal cellulases that extract sugars from tree cellulose differ from bacterial cellulases produced on the forest and ocean floors under different conditions.Submitted by DOE's Oak Ridge National Laboratory |