David Miller is an unassuming man. He maintains a hint of boyish charm—a self-effacing grin and glasses propped on the bridge of his nose. To look at him, one would never suspect that he stands at the nexus of an alliance that sprawls across the entire continental U.S.: the Carbon Capture Simulation Initiative (CCSI)—a vast web with personnel in eight states representing five Department of Energy laboratories, five esteemed universities, and an advisory board with over 20 industry partners who have hitched on for the ride. The coalition is unique, in both structure and mission, and David Miller, the technical director, is at its heart.
The initiative was launched in 2011 with a deceptively simple sounding objective: accelerate the development and deployment of carbon capture technologies by designing computational modeling tools that will speed development and scale-up. Carbon capture technologies are exactly what they sound like—technology that is designed to be incorporated into fossil-fueled power plants and isolate the CO2 before it can be released into the atmosphere. The current U.S. strategy for climate change mitigation relies on developing effective carbon capture methods as an integral component of the plan.
Full Story
Feature
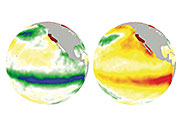
The weather patterns El Nino and La Nina may bring some ornery conditions to California later this century.
A research team from DOE's Pacific Northwest National Laboratory and Utah State University, funded by DOE ’s Office of Science, studied the patterns to gain a better sense of California’s future weather as the earth warms up. The scientists concluded that El Nino and La Nina, which alternately warm and cool the Pacific Ocean, could lead to at least a doubling of extreme droughts and floods in the state.
In the project, the research team looked at what happens to California in global climate models. To examine the most extreme case, the scientists simulated 2006 to 2080 using conditions in which very few efforts are made to reduce greenhouse gas emissions.
Full Story
See also…
 An ultra-high-resolution technique used for the first time to study polymer fibers that trap uranium in seawater may cause researchers to rethink the best methods to harvest this potential fuel for nuclear reactors.
An ultra-high-resolution technique used for the first time to study polymer fibers that trap uranium in seawater may cause researchers to rethink the best methods to harvest this potential fuel for nuclear reactors. Physicists at DOE’s Princeton Plasma Physics Laboratory (PPPL) are proposing a new way to process nuclear waste that uses a plasma-based centrifuge. Known as plasma mass filtering, the new mass separation techniques would supplement chemical techniques. It is hoped that this combined approach would reduce both the cost of nuclear waste disposal and the amount of byproducts produced during the process.This work was supported by PPPL's Laboratory Directed Research and Development Program.
Physicists at DOE’s Princeton Plasma Physics Laboratory (PPPL) are proposing a new way to process nuclear waste that uses a plasma-based centrifuge. Known as plasma mass filtering, the new mass separation techniques would supplement chemical techniques. It is hoped that this combined approach would reduce both the cost of nuclear waste disposal and the amount of byproducts produced during the process.This work was supported by PPPL's Laboratory Directed Research and Development Program. Government agencies, along with state and local governments, could receive a helping hand from a computer network security tool developed by computer scientists and engineers at DOE's Lawrence Livermore National Laboratory.
Government agencies, along with state and local governments, could receive a helping hand from a computer network security tool developed by computer scientists and engineers at DOE's Lawrence Livermore National Laboratory.  The newest addition to DOE’s National Renewable Energy Laboratory (NREL) is an experimental environmental chamber capable of making its occupants shiver or sweat.
The newest addition to DOE’s National Renewable Energy Laboratory (NREL) is an experimental environmental chamber capable of making its occupants shiver or sweat.